 The news follows after Boston Scientific was able to further extend its pain mitigation efforts, acquiring Intercept Intraosseous Nerve Ablation System developer, Relievant Medsystems for an upfront cash payment of $850m.Įlsewhere in the field of neurostimulation, Dutch competitor Onward Medical has started a Netherlands-based clinical feasibility study to assess its ARC-IM neurostimulator’s ability to improve haemodynamic instability in spinal cord injury patients. Background: The number of spinal cord stimulator (SCS) units sold in the United States (US) for the treatment of chronic pain has increased with a corresponding expansion in the number of different SCS platforms available. The market is largely dominated by a back-and-forth between Boston Scientific and its US-based competitor, Medtronic. GlobalData’s Medical Device Centre details how there are as many as 79 spinal cord stimulator devices currently in the development pipeline, with 54 of those in clinical development. 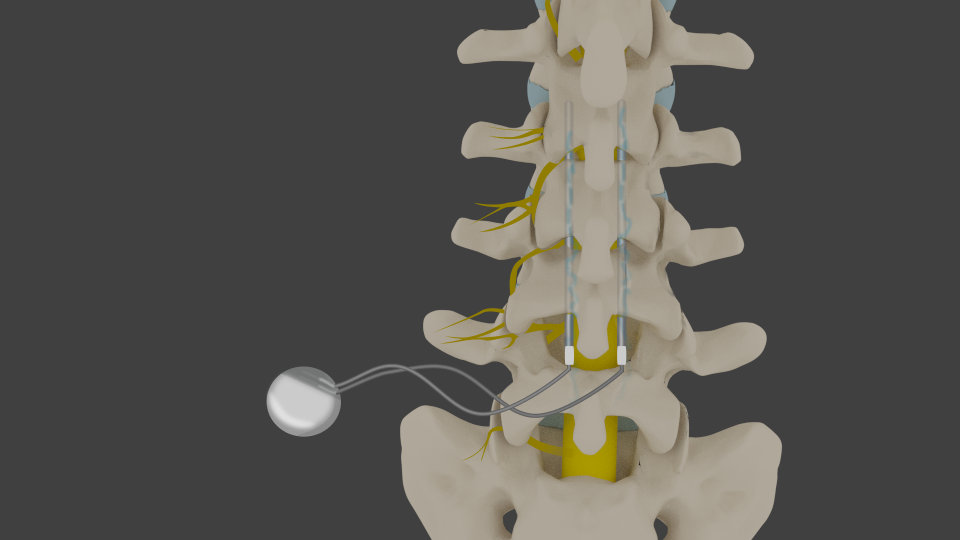 
“Backed by consistently strong clinical evidence, our transformative pain management solutions help individualize care and improve the quality of life for the many people living with chronic pain today.”īoston Scientfic’s study: Global, multicenter study of prospectively-enrolled patients utilizing SCS for Painful-Diabetic Peripheral Neuropathy, itself saw 81% of patients similarly reporting a 50% drop in pain.Īt the same time the company’s FAST Therapy study, a multiple prospective and real-world study taking place across Europe and the United States similarly saw 88% of SCS patients reporting 50% or greater pain relief. Clinical studies have shown that BurstDR technology delivers superior pain relief as compared to tonic stimulation 3, improves people's day-to-day. Jim Cassidy, president of neuromodulation at Boston Scientific, said: “Continued positive results from the SOLIS study illustrate the need for early and effective pain treatment when just the standard of care is not enough. BurstDR therapy is Abbott's exclusive stimulation technology that delivers pulses or bursts of mild electrical energy to alter pain signals as they travel from the spinal cord to the brain. The results were announced as part of the North American Neuromodulation Society Meeting (NANS) in Las Vegas and were released alongside updates on two other studies being carried out using Boston Scientific’s SCS system in different indications.ĭata from the trial showed that 85% of patients who declared a drop in pain reported a greater ability to take part in daily activities and a 25-point improvement in disability as measured by the Oswestry Disability Index. 
One-year data from the SOLIS trial demonstrates that Boston Scientific’s WaveWriter Alpha Spinal Cord Stimulation (SCS) system can reduce pain in chronic pain patients by 50%. Abbotts DISTINCT study, which enrolled 270 participants who suffered for an average of 12.8 years with pain, is the largest randomized controlled trial for SCS in people with chronic back pain. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
